Remote Clinical Trial Monitoring Tools Market Size and Growth Forecast 2026-2030
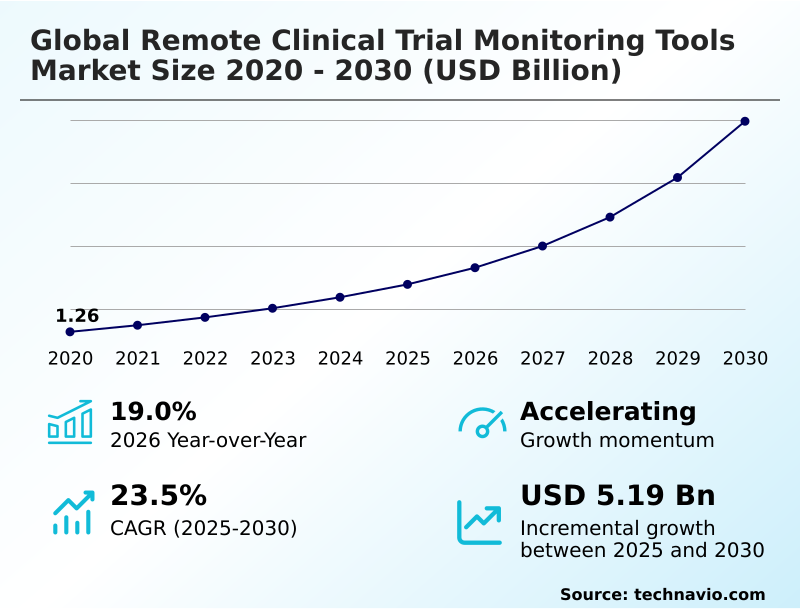
The Remote Clinical Trial Monitoring Tools Market size was valued at USD 2.77 billion in 2025 growing at a CAGR of 23.5% during the forecast period 2026-2030.
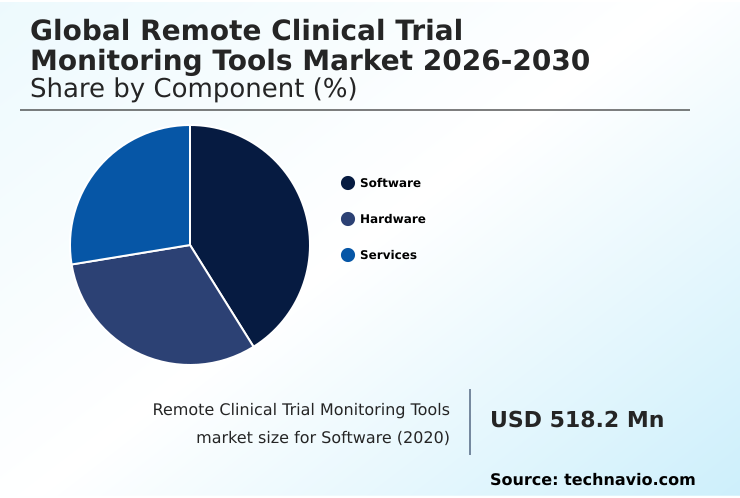
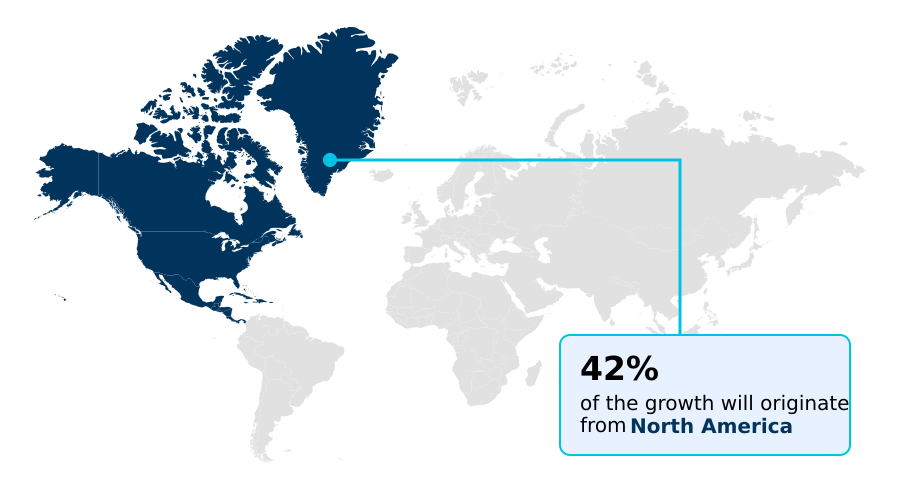
North America accounts for 41.7% of incremental growth during the forecast period. The Software segment by Component was valued at USD 1.00 billion in 2024, while the Pharmaceutical and biotech companies segment holds the largest revenue share by End-user.
The market is projected to grow by USD 6.70 billion from 2020 to 2030, with USD 5.19 billion of the growth expected during the forecast period of 2025 to 2030.
Get Key Insights on Market Forecast (PDF) Request Free Sample
Remote Clinical Trial Monitoring Tools Market Overview
The remote clinical trial monitoring tools market is defined by a strategic industry transition from site-centric oversight to technology-enabled, centralized methodologies. This evolution is driven by the need for greater efficiency in clinical development, where risk-based monitoring and remote source data verification have become standard practice. A key factor is the integration of electronic data capture systems with data from wearable biosensors and patient-facing mobile applications, creating a continuous stream of information. For instance, a contract research organization managing a multi-regional oncology trial utilizes a unified platform to perform remote data review, analyzing real-time digital biomarkers to detect adverse events. This proactive approach, compliant with FDA digital health guidelines, allows monitors to manage trial integrity without physical site visits. With North America contributing over 41% of the incremental growth, the region’s advanced digital maturity and supportive regulatory environment set the pace for global adoption, pushing vendors to innovate in areas like real-time site performance tracking and automated data reconciliation.
Drivers, Trends, and Challenges in the Remote Clinical Trial Monitoring Tools Market
The strategic adoption of remote clinical trial monitoring tools is increasingly influenced by the total cost of ownership, compelling decision-makers to evaluate how integrating EDC with remote monitoring tools impacts operational budgets.
While initial capital expenditure for platforms with advanced AI algorithms for clinical data anomaly detection can be significant, the long-term return on investment is realized through reduced site visit frequency and expedited data-lock timelines.
A key consideration is compliance with evolving regulatory frameworks, such as the technical standards for remote monitoring software validation outlined in the European Medicines Agency's reflection paper on AI. In a practical scenario, a CRO implementing a platform for automating remote source data verification for a large-scale cardiovascular study can reduce monitoring costs by over 30% compared to traditional methods.
This efficiency is crucial for managing the complexities of decentralized study participant management and ensuring adherence to Good Clinical Practice (GCP) guidelines. The benefits of using predictive analytics for clinical trial risk assessment further justify the investment, as these systems provide early warnings on site performance issues, allowing for proactive intervention and safeguarding trial integrity.
Primary Growth Driver: The increasing adoption of decentralized and hybrid clinical trial models is a primary driver for the expansion of the market.
The expansion of the market is primarily driven by the increasing industry adoption of decentralized and hybrid clinical trial models.
This strategic shift necessitates robust remote monitoring solutions to ensure data integrity and patient safety without requiring physical oversight at geographically dispersed sites.
Regulatory bodies, including the FDA, have bolstered this driver by issuing guidance that encourages risk-based approaches, for which remote monitoring tools are essential. These platforms provide the analytical capabilities to identify data anomalies and site performance issues in real time.
Furthermore, technological advancements in AI and cloud computing enable the automation of data cleaning and verification processes.
The proliferation of IoT devices and high-speed connectivity facilitates the seamless transmission of large data sets from sources like electronic health records, which are then managed by centralized monitoring platforms.
Emerging Market Trend: A primary trend is the market's shift from reactive to proactive oversight through the integration of predictive analytics and machine learning in monitoring tools.
The market is evolving from descriptive oversight to proactive intervention through the integration of predictive analytics and machine learning models. Modern platforms now utilize neural networks and advanced algorithms to analyze historical trial data, identifying patterns that indicate a high probability of future non-compliance or data anomalies. This shift allows for a more effective risk-based monitoring approach, optimizing resource allocation.
The increasing use of medical-grade wearable devices provides a continuous stream of physiological data, which, when fed into these systems, enables the early detection of adverse events. This trend supports the broader industry move toward decentralized trials, improving patient retention and data quality.
The ongoing refinement of these predictive models represents a fundamental change in how monitors interact with trial data, paving the way for a more efficient, data-driven future.
Key Industry Challenge: The preservation of data security and mitigation of cybersecurity vulnerabilities represent a primary challenge for the market.
A significant challenge constraining the market is the complexity of ensuring data security and mitigating cybersecurity risks in decentralized trial environments. The transmission of sensitive protected health information across various digital networks creates numerous vulnerabilities. Organizations must invest heavily in advanced encryption and continuous monitoring systems to protect the trial ecosystem in compliance with regulations like HIPAA.
Another major barrier is the lack of standardization and system interoperability between disparate software systems, such as EDC platforms and electronic health records. This fragmentation often forces manual data reconciliation, increasing the risk of human error.
Additionally, inconsistent regulatory landscapes and varying local privacy laws, especially regarding cross-border data transfer and data sovereignty requirements, create significant logistical hurdles for sponsors conducting global trials.
Explore Full Market Dynamics Analysis Request Free Sample
Remote Clinical Trial Monitoring Tools Market Segmentation
The remote clinical trial monitoring tools industry research report provides comprehensive data including region-wise segment analysis, with forecasts and analysis for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
Component Segment Analysis
The software segment is estimated to witness significant growth during the forecast period.
The software segment is the architectural core of the remote clinical trial monitoring tools market, providing the essential electronic data capture systems and platforms for trial management.
This segment's growth is tied to the industry-wide adoption of cloud-based architecture and generative artificial intelligence to manage the data streams from wearable biosensors.
In practice, organizations leverage these solutions to automate data cleaning and verification, reducing manual error and ensuring the integrity of patient-reported outcomes for regulatory submissions.
The shift to decentralized trial models has intensified the demand for software that offers robust remote source data verification capabilities.
As of 2023, this segment accounted for a dominant share of the market, reflecting its criticality in enabling the operational efficiencies required for modern clinical development.
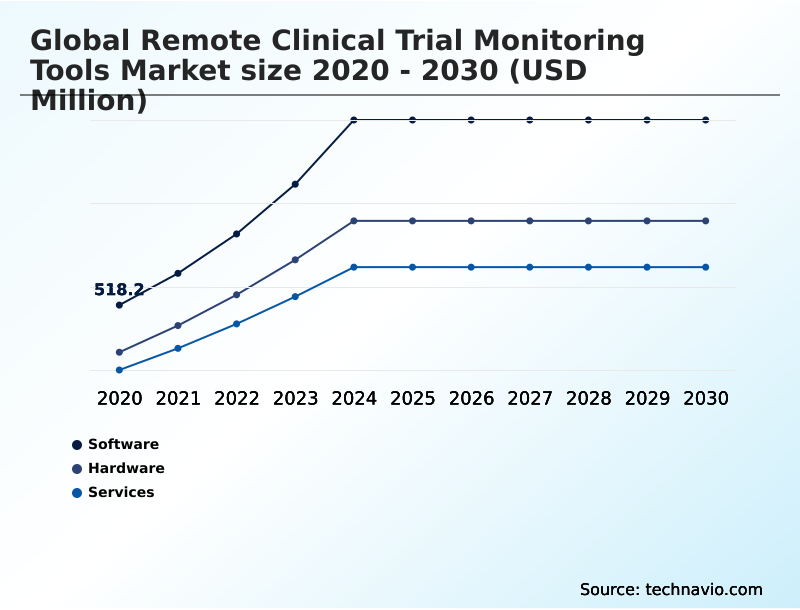
The Software segment was valued at USD 1.00 billion in 2024 and showed a gradual increase during the forecast period.
Remote Clinical Trial Monitoring Tools Market by Region: North America Leads with 41.7% Growth Share
North America is estimated to contribute 41.7% to the growth of the global market during the forecast period.
The geographic landscape is led by North America, which is projected to contribute approximately 41% of the market's growth, driven by a mature digital health ecosystem and supportive FDA policies promoting remote data acquisition.
In Europe, which accounts for over 24% of the market opportunity, adoption is shaped by the stringent requirements of the General Data Protection Regulation (GDPR), demanding advanced data anonymization features and secure cloud-based repositories.
Asia is the fastest-growing region, with countries like China implementing specific cybersecurity protocols for remote clinical monitoring data, compelling global technology providers to localize their data storage solutions.
Across all regions, the push for greater system interoperability is a constant, with a focus on standardizing remote source data verification protocols to streamline multi-regional clinical trials and reduce the administrative burden on investigative sites.
Customer Landscape Analysis for the Remote Clinical Trial Monitoring Tools Market
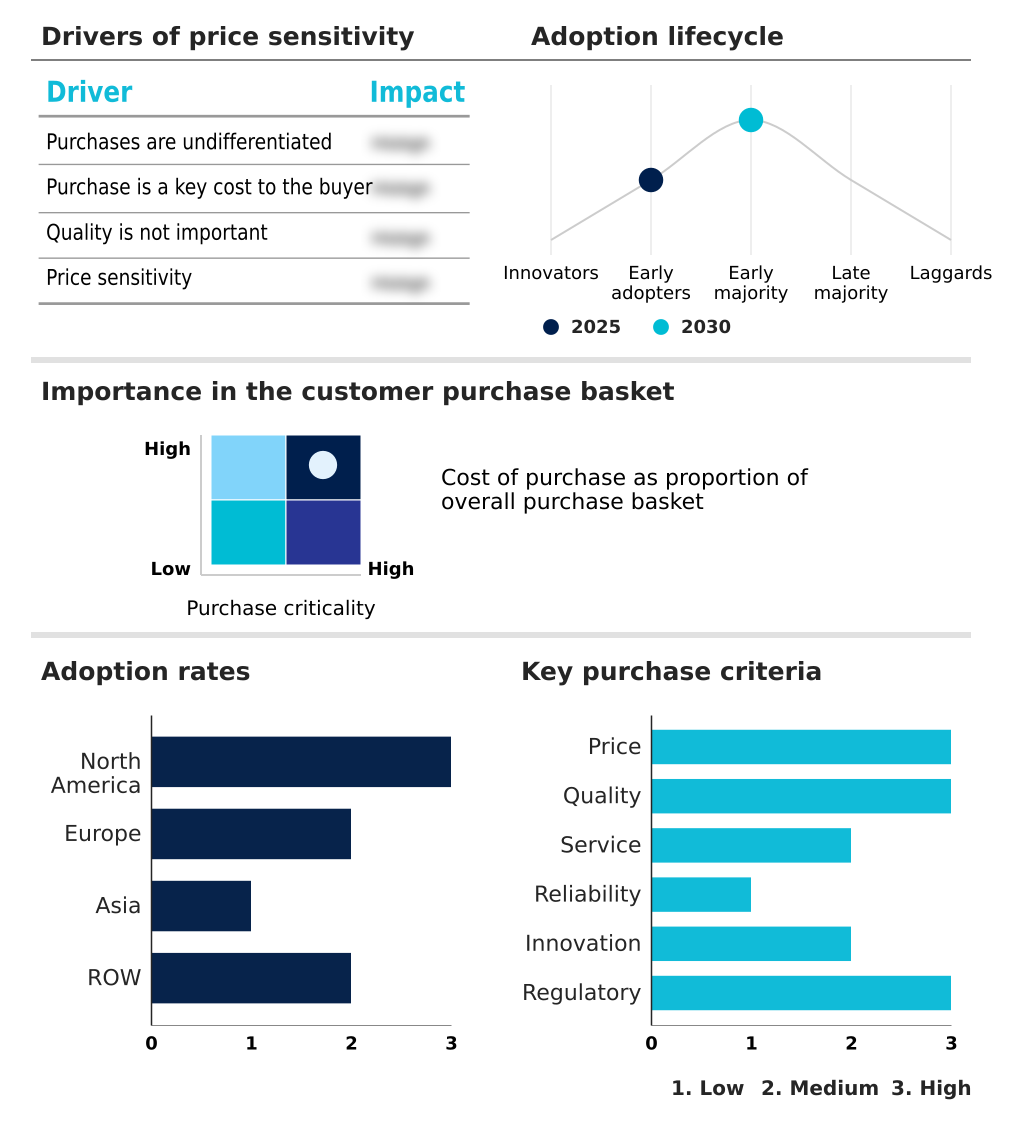
The remote clinical trial monitoring tools market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the remote clinical trial monitoring tools market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Competitive Landscape of the Remote Clinical Trial Monitoring Tools Market
Companies are implementing various strategies, such as strategic alliances, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the remote clinical trial monitoring tools market industry.
Advarra Inc. - Offerings include eRegulatory and electronic data capture platforms with remote monitoring capabilities, document management features, and tools for real-time site performance tracking for clinical trial sponsors.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Advarra Inc.
- Anju Software Inc.
- Castor Research Inc.
- Cloudbyz Inc.
- Ennov SAS
- Florence Healthcare.
- IQVIA Holdings Inc.
- Medable Inc.
- Medidata
- Medrio Inc.
- Merative L.P.
- Microsoft Corp.
- Obvio Health USA Inc.
- OpenClinica LLC
- Oracle Corp.
- Salesforce Inc.
- SAP SE
- Signant Health
- Veeva Systems Inc.
- Zoom Video Communications Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Developments in the Remote Clinical Trial Monitoring Tools Market
- In August 2025, the European Medicines Agency published a comprehensive reflection paper on using artificial intelligence in remote clinical trial monitoring, providing clear technical standards for software validation.
- In October 2025, a leading technology provider specialized in clinical software introduced a machine learning module utilizing natural language processing to automate the reconciliation of remote source data.
- In November 2025, an international regulatory consortium finalized new technical requirements for the interoperability of remote monitoring tools in multi-regional clinical trials, establishing a common architecture for secure data exchange.
- In March 2025, a leading global provider of clinical trial software introduced a specialized analytics module that employs neural networks to predict site-level enrollment delays and potential protocol deviations.
Research Analyst Overview: Remote Clinical Trial Monitoring Tools Market
The remote clinical trial monitoring tools market is advancing beyond simple data collection toward intelligent, predictive oversight. Boardroom decisions on technology procurement are now heavily influenced by a platform's ability to integrate machine learning models for proactive risk detection, a capability that directly impacts trial timelines and budget adherence.
The adoption of risk-based monitoring frameworks, championed by bodies like the International Council for Harmonisation (ICH), necessitates tools with sophisticated analytical capabilities. We observe that platforms incorporating natural language processing to automate data reconciliation are gaining a competitive edge.
This technological shift is a response to the operational complexities of managing decentralized clinical trials and the sheer volume of data generated by wearable biosensors and other IoT devices. North America's market dominance, contributing over 41% to growth, underscores the value placed on these advanced features in a mature regulatory environment.
The focus is now on achieving seamless system interoperability and deploying generative AI to transform data verification workflows.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Remote Clinical Trial Monitoring Tools Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 293 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 23.5% |
| Market growth 2026-2030 | USD 5185.6 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 19.0% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, The Netherlands, Spain, Russia, China, India, Japan, South Korea, Indonesia, Thailand, Singapore, Australia, UAE, Brazil, South Africa, Saudi Arabia and Turkey |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Remote Clinical Trial Monitoring Tools Market: Key Questions Answered in This Report
-
What is the expected growth of the Remote Clinical Trial Monitoring Tools Market between 2026 and 2030?
-
The Remote Clinical Trial Monitoring Tools Market is expected to grow by USD 5.19 billion during 2026-2030, registering a CAGR of 23.5%. Year-over-year growth in 2026 is estimated at 19.0%%. This acceleration is shaped by rise of decentralized and hybrid clinical trial models, which is intensifying demand across multiple end-use verticals covered in the report.
-
-
What segmentation does the market report cover?
-
The report is segmented by Component (Software, Hardware, and Services), End-user (Pharmaceutical and biotech companies, Contract research organizations, Healthcare IT and service providers, and Academic and research institutes), Deployment (Cloud based, and On premises) and Geography (North America, Europe, Asia, Rest of World (ROW)). Among these, the Software segment is estimated to witness significant growth during the forecast period, driven by rising adoption across key application areas. Each segment includes detailed qualitative and quantitative analysis, along with historical data from 2020-2024 and forecasts through 2030 with year-over-year growth rates.
-
-
Which regions are analyzed in the report?
-
The report covers North America, Europe, Asia and Rest of World (ROW). North America is estimated to contribute 41.7% to market growth during the forecast period. Country-level analysis includes US, Canada, Mexico, Germany, UK, France, Italy, The Netherlands, Spain, Russia, China, India, Japan, South Korea, Indonesia, Thailand, Singapore, Australia, UAE, Brazil, South Africa, Saudi Arabia and Turkey, with dedicated market size tables and year-over-year growth for each.
-
-
What are the key growth drivers and market challenges?
-
The primary driver is rise of decentralized and hybrid clinical trial models, which is accelerating investment and industry demand. The main challenge is data security and cybersecurity vulnerabilities, creating operational barriers for key market participants. The report quantifies the impact of each driver and challenge across 2026 and 2030 with comparative analysis.
-
-
Who are the major players in the Remote Clinical Trial Monitoring Tools Market?
-
Key vendors include Advarra Inc., Anju Software Inc., Castor Research Inc., Cloudbyz Inc., Ennov SAS, Florence Healthcare., IQVIA Holdings Inc., Medable Inc., Medidata, Medrio Inc., Merative L.P., Microsoft Corp., Obvio Health USA Inc., OpenClinica LLC, Oracle Corp., Salesforce Inc., SAP SE, Signant Health, Veeva Systems Inc. and Zoom Video Communications Inc.. The report provides qualitative and quantitative analysis categorizing companies as dominant, leading, strong, tentative, and weak based on their market positioning. Company profiles include business segment analysis, SWOT assessment, key offerings, and recent strategic developments.
-
Remote Clinical Trial Monitoring Tools Market Research Insights
The market's dynamics are shaped by procurement decision-makers prioritizing integrated platforms over fragmented point solutions to manage decentralized clinical trials. The increasing reliance on real-time data from wearable technology necessitates robust back-end systems that can handle high-frequency data streams for continuous patient monitoring.
In practice, a sponsor must ensure its chosen clinical monitoring platform adheres to the EU's Clinical Trials Regulation (CTR 536/2014) for multi-regional data exchange, a key vendor qualification criterion. This regulatory pressure, combined with the operational need to streamline data reconciliation workflows, drives investment in tools offering advanced analytics and automated query management.
The software segment's dominance highlights the strategic importance of scalable, cloud-based architectures in modernizing clinical oversight and ensuring data integrity across global study sites.
We can help! Our analysts can customize this remote clinical trial monitoring tools market research report to meet your requirements.


 RIA -
RIA - 
